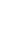-

 EMD
EMDEnvironmental Materials Development Laboratory in Hanyang University
Research
The Environmental Materials Development Laboratory focuses on producing useful materials and developing environmental materials using waste.
more view


-
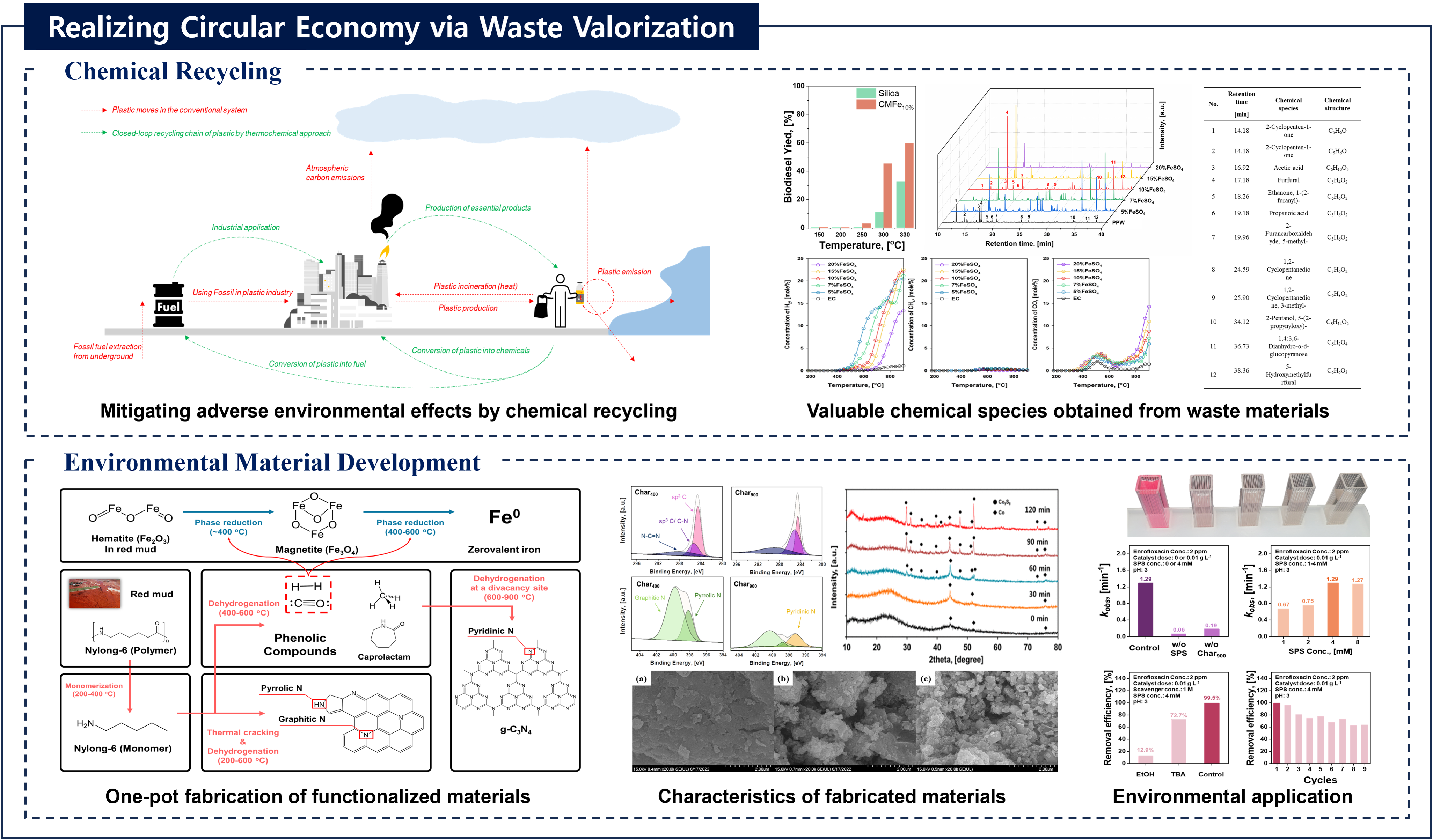
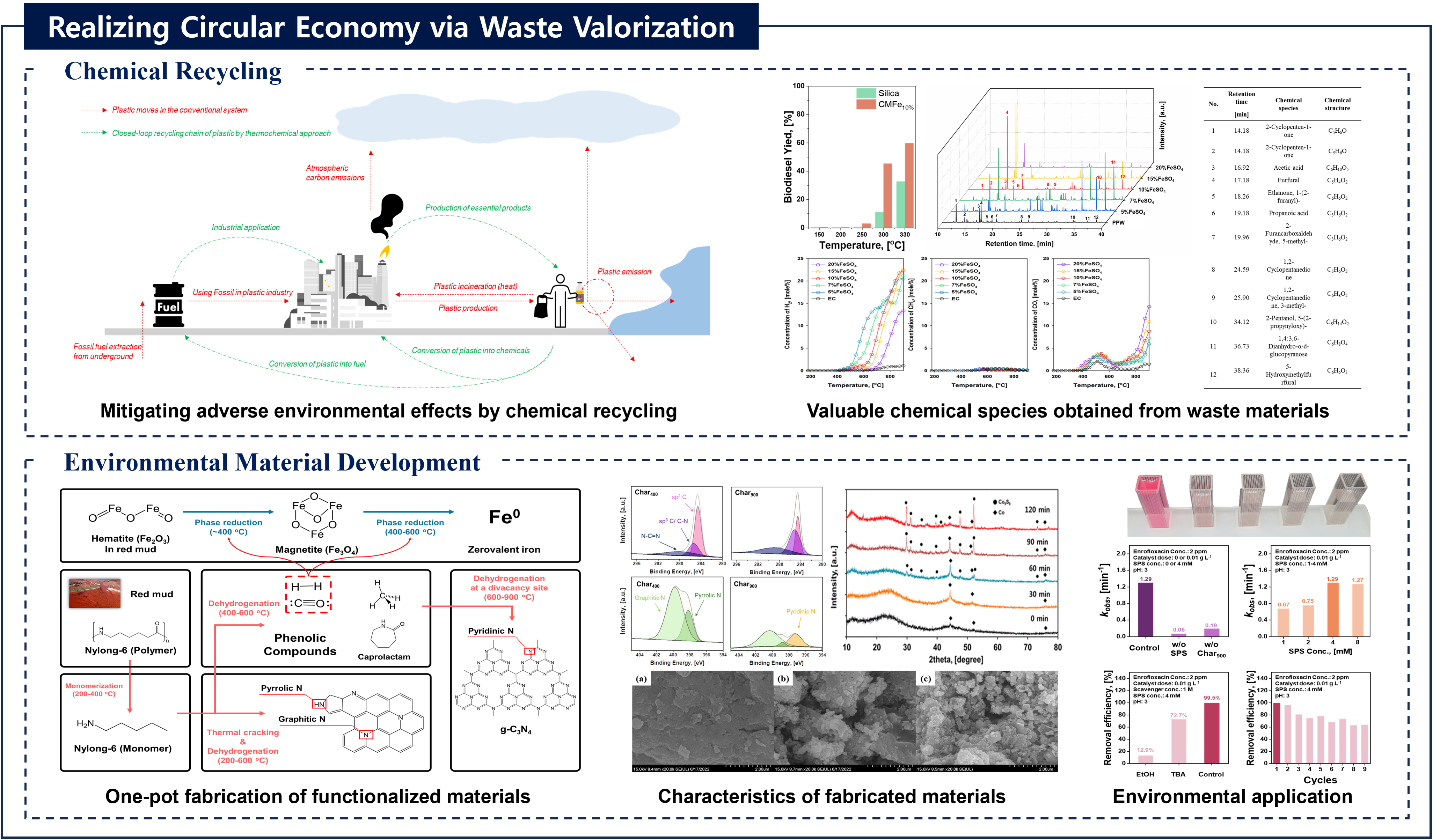
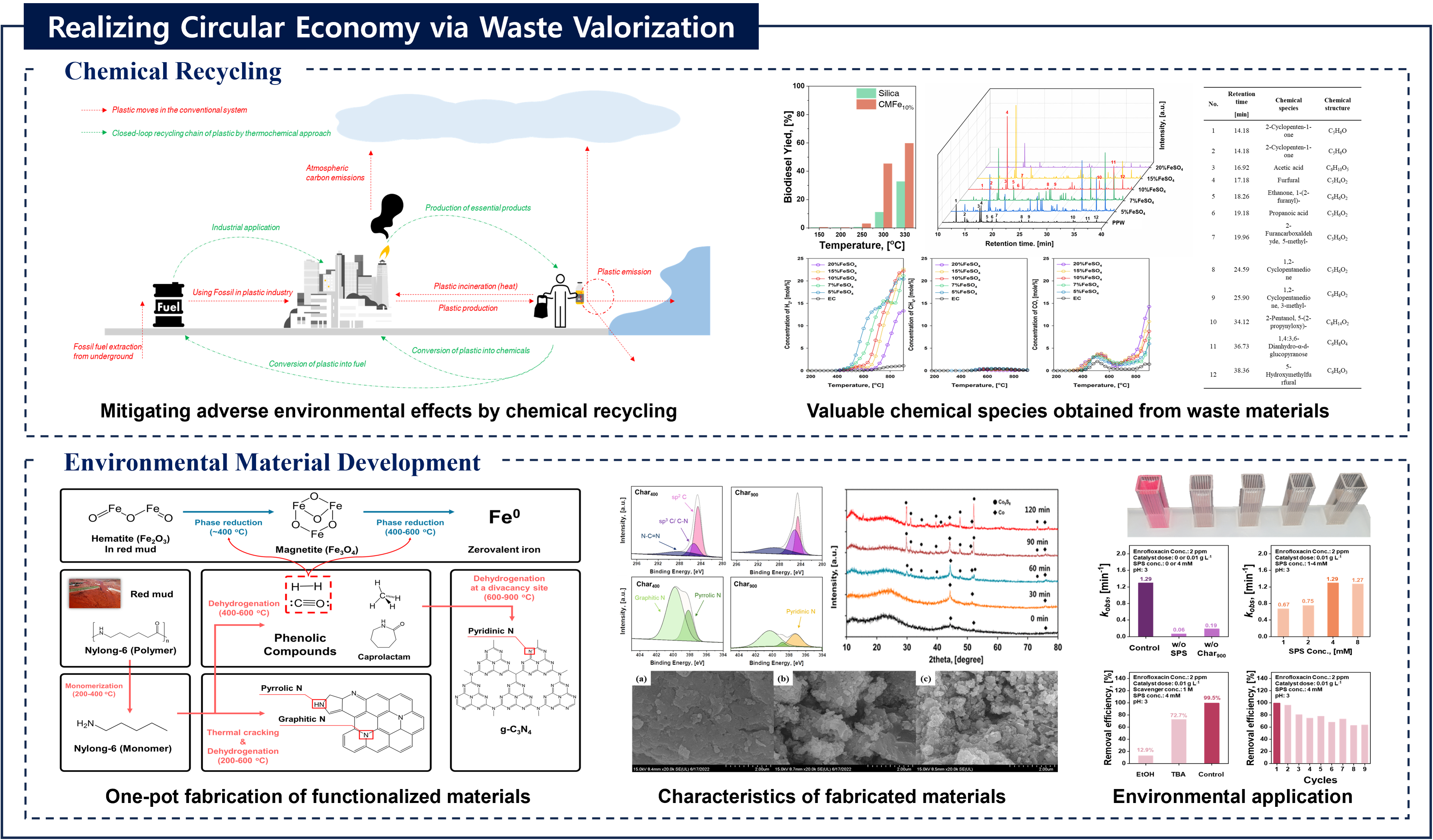
Recovery of chemicals and energy through thermo-chemical processing of plastic waste
Journal: Recovery of chemicals and energy through thermo-chemical processing of plastic wasteAuthors: Taewoo Lee, Dohee Kwon, Sangyoon Lee, Youkwan Kim, Jee Young Kim, Hocheol Song, Sungyup Jung, Jechan Lee, Yiu Fai Tsang, Ki-Hyun Kim, Eilhann E KwonAbstract:To mitigate the various socioeconomic/environmental consequences associated with plastic waste, it is crucial to adopt strategic measures aimed at source reduction. In this regard, the thermo-chemical approach is a promising technical option to realize this objective within the framework of the circular economy. Such approach involves transforming plastic waste into chemicals/fuels, which contributes to the build-up of a more sustainable and resource-efficient platform. Precise control over yield and selectivity towards target chemicals (monomers, light olefins, and benzene, toluene, ethylbenzene, and xylene isomers (BTEXs)) and fuels (transportation fuels and syngas) is achievable by manipulating operating parameters for the thermo-chemical platform despite the possibly marked influence of the waste composition on product distribution. This review aims to delineate a technically viable pathway of the thermo-chemical approach with the discussion on the physico-chemical properties and compositional characteristics of plastics, technical alternatives for their recycling, and the associated environmental risks (improper disposal practices including mismanagement, landfilling, and incineration). This review helps open a new path for the development of a strategic technical approach within thermo-chemical processing to integrate different facets of plastic waste recycling. Thus, it will contribute to the realization of a closed-loop circular economy within the plastic value chain by focusing on thermo-chemical recycling of plastic waste.Keywords: Circular economy; Sustainability; Waste valorization; Thermo-chemical process; Plastic wasteDOI: https://doi.org/10.1016/j.pecs.2025.101219
2025-02-17 -
CO2-Mediated Pyrolysis for Syngas Generation from Wood-Plastic Composite Waste
Journal: CO2-Mediated Pyrolysis for Syngas Generation from Wood-Plastic Composite WasteAuthors: Dohee Kwon, Jee Young Kim, Hocheol Song, Jaewon Lee, Yiu Fai Tsang, Eilhann E KwonAbstract:Wood-plastic composites (WPCs) are extensively utilized in construction, furniture, and outdoor applications due to their superior physicochemical properties. However, their low biodegradability presents a critical challenge for waste management. This study explores upcycling WPC waste into syngas via CO2-mediated pyrolysis. CO2 serves as reaction medium, enhancing thermal decomposition and partial oxidation of volatile matters (VMs) generated from pyrolysis of WPCs to form carbon monoxide (CO) through homogeneous reactions. To further optimize the role of CO2, a multi-stage pyrolysis operating isothermally at 700 ˚C was employed. In this configuration, the reactivity of CO2 was more pronounced at lower temperatures than that during single-stage pyrolysis, indicating that the additional thermal energy accelerated CO2-induced reactions. The introduction of a nickel-based catalyst accelerated the CO2-induced homogeneous reactions by facilitating the adsorption and reaction of CO2 with VMs during pyrolysis. Thus, utilizing CO2 as a reaction medium in the catalytic pyrolysis process represents a strategic pathway for the upcycling of WPC waste.Keywords: Wood plastic composite; Waste-to-Energy; Pyrolysis; Syngas; Carbon dioxideDOI: https://doi.org/10.1016/j.energy.2025.134917
2025-02-17 -
Development of ceria-supported metal-oxide (MOx/CeO2) catalysts via a one-pot chemical vapor deposit…
Journal: Development of ceria-supported metal-oxide (MOx/CeO2) catalysts via a one-pot chemical vapor deposition (OP-CVD) technique: Structure and reverse water gas shift reaction studyAuthors: Amol Pophali, Ryuichi Shimogawa, Lihua Zhang, Gihan Kwon, Kwangsuk Yoon, Jangeon Roh, Do Heui Kim, Hocheol Song, Anatoly I Frenkel, Taejin KimAbstract:Current synthesis techniques for metal oxide (MOx)-supported catalysts have certain limitations of undesired target loading, ineffective dispersion of active species over the surface, uncontrolled particle size of active species, and complicated synthesis steps. We developed a one-pot chemical vapor deposition (OP-CVD) methodology; by using which a solid metal precursor forms a vapor in a controlled condition and gets supported over the surrounding matrix. The theoretical stability followed by experimental validation using TGA is crucial for selecting the metal precursors. Three simple steps viz. premixing, dispersion, and rapid fixation by calcination are involved in the catalyst development via the OP-CVD approach. This study solely focused on the synthesis of 3d transition MOx over ceria support. The physicochemical characterizations of the prepared catalysts were performed by XRD, ICP-OES, SEM-EDX, CO pulse chemisorption, XANES, and EXAFS analyses to understand the crystal structure of involved species, target metal loading, dispersion, and particle size and prove the feasibility and viability of OP-CVD. The prepared catalysts were further tested for reverse water gas shift (RWGS) reaction to link their structural information with activity. The RWGS reaction data showed that the CO activity and CO selectivity were metal - and metal precursor-dependent. Higher CO activity of > 0.1 mol/h g-cat was observed for Cu and Co-based catalysts, with CO selectivity of ∼100 %. This study provides an opportunity to produce efficient supported catalysts in a convenient way, providing effective catalytic activity.Keywords: Catalysts; One-pot chemical vapor deposition; Organometallic precursor; Monomer; Reverse water gas shift reaction; CeriaDOI: https://doi.org/10.1016/j.cej.2024.158726
2025-02-17






News
more


-
23
2024.12송호철 교수, 환경 재료 개발로 폐기물 순환 경제를 실현하는 세계 상위 0.1%…2024 세계 최상위 0.1% 연구자 선정, 송호철 자원환경공학과 교수폐기물을 활용한 환경 재료 개발"한양인이 스스로의 가능성을 믿고, 꾸준히 도전했으면"'2024 세계에서 가장 영향력 있는 연구자 HCR(Highly Cited Researchers, 이하 HCR)'에 송호철 자원환경공학과 교수가 이름을 올렸다. HCR은 글로벌 조사분석기업 클래리베이트(Clarivate)가 과학 및 사회과학의 21개 특정 분야, 여러 영역을 다루는 교차 분야에서 선정한 전 세계에서 가장 영향력 있는 연구자이다. 송 교수를 포함해 한양대 교수 7인이 지난해 HCR에 선정됐다.송 교수는 여러 분야에 걸쳐 많이 인용된 논문을 뜻하는 '크로스 필드(Cross-Field)' 분야의 상위 0.1% 연구자로 이름을 올렸다. 그는 2020년부터 5년 연속으로 정보 분석 기업 엘스비어(Elsevier)가 선정한 세계 상위 2% 연구자에 이름을 올리고 있다.송 교수는 폐기물·환경 분야의 혁신적인 연구성과 창출의 공로로 ‘과학기술훈장 도약장’과 환경부 장관상을 수상하며 연구 분야를 선도하고 있다. 폐기물 기반의 환경 재료를 개발하며 환경 보호에 힘쓰는 송 교수를 만나 자세한 이야기를 나눴다. ▲ '세계에서 가장 영향력 있는 연구자 HCR(Highly Cited Researchers)'은 과학 및 사회과학, 교차 분야서 지난 10년간 출판된 논문 중 인용 빈도가 가장 높은 논문을 작성한 세계 상위 0.1% 연구자를 의미한다.| 세계 상위 0.1% 연구자 선정, 빛나는 여정의 증명'2024년 세계 상위 0.1% 연구자(HCR)'에 선정되셨어요. 소감 부탁드립니다. 전혀 예상하지 못 했어요. 선정됐다는 소식을 들으니 기분이 정말 좋았습니다. 제가 해왔던 연구들이 다른 연구자들에게 도움과 흥미를 줄 수 있다는 것을 인정받은 것 같아요. 학술적 가치를 인정 받은 그동안의 연구들에 큰 보람을 느낍니다. 세계 상위 2% 연구자에도 5년 연속 꾸준히 이름을 올리고 계십니다. 그동안 교수님께서 집중해 오신 연구를 소개해 주신다면요.기존 연구는 환경 재료가 아니었어요. 토양이나 지하수에 있는 오염을 평가하고, 오염물질을 제거하는 연구를 주로 해왔죠.그러나 최근 10여년 전부터 '환경 재료'에 관심이 생겨 본격적으로 연구하기 시작했습니다. 우리가 주로 폐기물이라고 일컫는 물질들, 예를 들어 농축산 폐기물·산업 폐기물·생활 폐기물 등을 원료로 활용해 열화학 공정을 거쳐 탄소 기반의 물질을 만들고 있어요. 해당 물질들은 환경 내 존재하는 다양한 오염 물질을 처리하는 데 사용되고 있고요. | 폐기물을 활용한 새로운 환경 재료 개발'환경 재료' 라는 것이 구체적으로 무엇인가요. 환경 재료는 물속이나 토양 내에 있는 오염물질을 제거하는 목적을 지닌 물질들을 의미합니다. 흡착하거나 산화하는 과정을 통해 빠른 시간 안에 오염물질을 저감하는 것이죠. 환경 재료는 종류가 매우 다양하고, 우리가 적용할 수 있는 분야가 넓어요. 특히 폐기물을 활용한 환경 재료 연구는 기존에 논문화하기 어려운 연구의 한계를 넘어 다양한 재료를 활용할 수 있다는 점에서 합리적입니다.폐기물은 경제성 확보하는 차원에서 매우 유용해요. 폐기물은 버리는 물질이잖아요. 버리는 물질을 원료로 활용해 우리가 활용할 수 있는 새로운 물질을 만든다는 점에서 큰 의미를 지니죠. '폐기물'을 활용해 유용 전구 물질을 생산한다고 하셨습니다. 해당 물질은 무엇이며, 어떻게 환경오염 저감에 기여하나요. 폐기물이 여러 공정 단계를 거치면 몇 가지 부산물이 나와요. 먼저, 가스와 타르라고 불리는 액상 물질이 나옵니다. 또한 우리가 환경 재료로 활용하는 고체 물질도 있습니다. 유용 전구 물질은 액상에 포함된 화학 물질입니다. 유용 전구 물질은 조건만 잘 맞추면 화학 산업에서 원료 물질로 활용할 수 있어요. 폐기물 공정 과정에서 큰 비용이 드는 합성 방법이 아닌, 추출 방법으로 유용한 물질을 활용할 수 있게 되는 것이죠. 교수님이 지향하시는 '폐기물 순환 경제'는 어떤 모습인가요. 생활 속에서 폐기물은 생겨날 수밖에 없어요. 그러나 문제는, 폐기물의 처리 과정에서 높은 비용과 극심한 오염이 발생하게 된다는 것이죠. '여러 공정을 통한 폐기물을 다른 물질로 전환할 수 있다면', '전환 과정에서 에너지를 얻고, 산업에 필요한 유용 전구 물질을 생산할 수 있다면', '오염 물질을 저감할 수 있는 환경 재료까지 만들 수 있다면 얼마나 환경적일까'라는 생각을 합니다. 오염 저감과 동시에 부가가치 창출도 가능하니까요. 이런 순환 경제를 꼭 실현하고 싶어요. ▲ 송 교수는 폐기물 기반 환경 재료 개발 및 연구를 통한 순환경제 실현에 기여한 공을 인정받아 '세계에서 가장 영향력 있는 연구자 HCR(Highly Cited Researchers)'에 선정됐다. ⓒ 신문정 기자연구하시면서 발견하신 새로운 접근법이나 기술이 있다면요. 제 연구는 '공동 열분해'라는 과정을 거치게 돼요. 공동 열분해 과정이란 유기성 폐기물을 무기성 산업 폐기물이나 금속을 포함하는 산업 폐기물과 함께 열분해하는 과정을 말하죠. 해당 과정을 거쳐 만들어진 새로운 환경 재료는 촉매 특성을 띠게 돼요.기존의 탄소 유래 물질은 흡착 특성이 강한 반면, 촉매 특성은 떨어지거든요. 그러나 공동 열분해 과정을 거치면 강한 흡착 특성도 가지는 동시에 촉매 특성도 유지할 수 있게 됩니다. 교수님이 생각하시는 환경 재료의 현주소는 어떤가요. 환경 재료와 관련한 연구는 아직 초기 단계에요. 학교나 연구소 등에서 여러 가지 연구가 수행되고 있지만, 대부분 실험실에서 이뤄지는 소규모 실험입니다. 실질적으로 환경 재료 연구가 실용화되거나 상용화되는 경우는 드물어요. 이런 부분을 보완해서 보다 적극적으로 연구를 이어나가야겠다는 생각을 합니다.우리가 살아가는 현재, 환경 재료 개발 및 연구가 필요한 가장 큰 이유는 '경제성'에 있어요. 여러 가지 환경 재료의 효율을 높이기 위해서는 그만큼의 비용이 들어갑니다. 효율성을 위해서 저비용의 물질 생산이 필요한데, 상당히 구현하기 어렵거든요. 이에 꾸준한 연구를 통해서 보다 경제적이고 효용성 있는 물질을 실질적으로 상용화하기 위해 연구하고 있습니다. 지금까지 연구를 해오시면서 가장 기억에 남는 순간은 언제였나요. 학생들과 함께 연구하는 순간이 가장 기억에 남아요. 특히 학생들과 논문을 작성하는 시간은 상당한 인고의 시간입니다. 함께 수정하고, 보완한 학생들의 논문은 저널에 투고하죠. 저널 심사에 따라 수정하는 작업도 굉장히 오래 걸려요. 해당 과정들이 어렵기도 하지만, 동시에 보람을 느끼는 때예요.가장 보람됐던 순간은 '한양대에 오는 것이 확정된 순간'이에요. 저도 한양대를 졸업했거든요. 다시 학교로 돌아올 수 있다는 사실에 큰 기쁨을 느꼈습니다. | "스스로가 똑똑하다는 것을 알고 나아갔으면"연구자로서 교수님의 원동력이 궁금합니다.지적 호기심과 함께 연구하는 순간이 원동력이 돼요. 중요한 주제에 대해 고민하고, 그 주제를 학생들과 함께 연구해 나가는 과정이 정말 값지거든요. 제가 가르쳤던 학생들이 좋은 연구자로 사회에 나가는 것을 볼 때면 보람을 느낍니다.제가 개발하고 있는 여러 환경 재료가 우리나라 산업에 많이 도입돼서 상용화되길 바라요. 그때까지 꾸준히 연구를 해나갈 생각입니다. ▲ 송 교수는 "학생들이 자신감을 가지고 새로운 것에 꾸준히 도전하며 능력을 키워갔으면 좋겠다"고 바람을 전했다. ⓒ 신문정 기자미래에 연구자가 될 한양인에게 격려 및 조언 한마디 부탁드립니다. 학생들이랑 같이 이야기하다 보면 우리 한양인이 정말 똑똑하다는 것을 느껴요. 그러나 정작 그들은 자신들이 얼마나 똑똑하고 창의적인지 잘 모르는 것 같아요. 자신들이 뛰어나다는 것을 알고, 자신감을 가졌으면 좋겠습니다. 자신감을 가지고 무엇이든 시도하다 보면, 자신도 모르던 새로운 능력을 발견할 수 있을 것이라 믿어요. 마지막으로 전하고 싶으신 말씀이 있다면요. 연구를 하면서 건설환경공학과 김기현 교수님, 자원환경공학과 권일한 교수님을 비롯한 많은 분의 도움을 받았습니다. 공동 연구뿐 아니라 인간적인 교류를 통해 많은 것을 배웠습니다. 제 연구에 도움을 주신 모든 분께 진심으로 감사드린다고 전하고 싶습니다. 출처 : 뉴스H(http://www.newshyu.com)
-
23
2024.12한양대 연구자 7인, 2024 세계 최상위 0.1% 연구자에 선정<폐기물 순환경제를 실현하는 환경공학의 선구자>자원환경공학과 송호철 교수송호철 자원환경공학과 교수는 크로스 필드(Cross-Field) 분야 상위 0.1% 연구자로 이름을 올렸다.송 교수는 폐기물 기반 자원화 및 환경재료 개발 연구를 주로 수행하고 있다. 농축산폐기물, 생활폐기물, 산업폐기물 등을 대체 자원으로 활용하여 산업에 필요한 에너지와 유용전구물질을 생산하고 환경 내 오염물질 저감을 위한 촉매나 흡착제를 개발하고 있다. 이러한 연구를 통해 폐기물의 처리 과정과 자원 공급 과정에서 발생하는 비용과 오염물질 배출을 줄이고 동시에 부가가치를 창출함으로써 폐기물 순환경제 구축에 선도적 역할을 하고 있다.한편 송 교수는 폐기물·환경 분야의 혁신적인 연구성과 창출의 공로로 '과학기술훈장 도약장' 과 환경부 장관상을 수상한 바 있다.출처 : 뉴스H(http://www.newshyu.com) (일부 발췌)
-
11
2024.05세종대 환경에너지융합학과 송호철 교수, 과학기술진흥 훈장 수상세종대학교(총장 배덕효) 환경에너지융합학과 송호철 교수(사진)는 지난 4월 21일 열린 ‘2021년 과학·정보 통신의 날 기념식'에서 과학기술진흥 훈장을 받았다.과학·정보 통신의 날 기념식은 제54회 과학의 날과 제66회 정보 통신의 날을 맞아 정보통신의 중요성과 의의를 높이고, 과학 정보통신인들에게 긍지를 심어주기 열렸다. 올해는 코로나19 확산 방지를 위해 온·오프라인으로 진행됐다.이날 행사에서는 과학기술·정보통신 진흥과 국가연구개발에 공헌한 유공자 총 151명에게 정부 포상이 수여됐다.송 교수는 “수상에 대해 기대를 전혀 하지 않았는데 그동안의 과학기술 진흥 연구성과를 인정받아 훈장을 받게 되어 매우 영광이다. 앞으로도 환경 분야와 환경 정화를 위한 재료 개발 연구를 꾸준히 할 계획이다”라고 말했다.송 교수는 2011년부터 세종대 환경에너지융합학과 교수로 재직하고 있으며, 오염 토양과 오염 지하수 정화제 개발 분야에서 전문적인 연구를 수행하고 있다.출처 : 서울시티(http://www.seoulcity.co.kr)
Gallery
more